
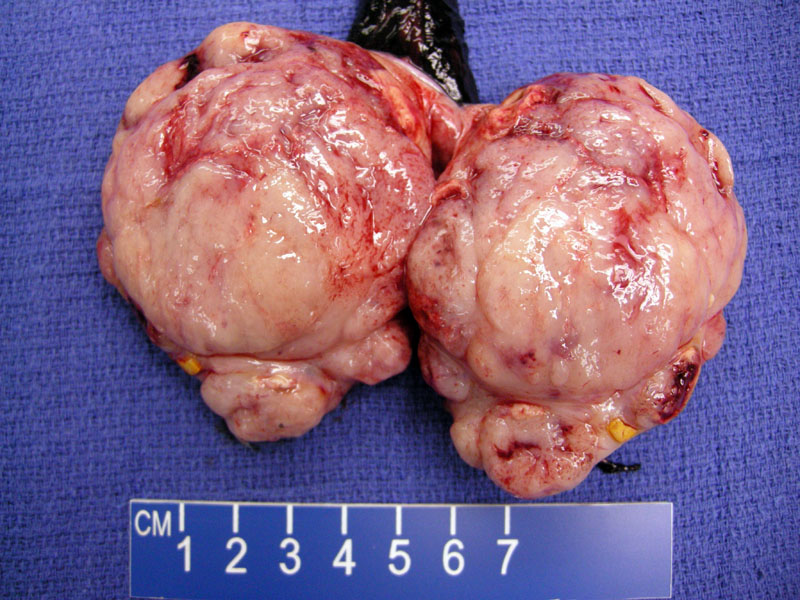
Grossly, the tumor is firm, bulky and lobulated, typically ranging 5-10 cm in diameter. The cut surface is gray-white and homogenous. Hemorrhage and necrosis is uncommon, but may be seen in larger tumors. The tumor is usually confined to the testes, but extension into the epididymis and spermatic cord is seen in 5-8% of cases.1
Another example of a seminoma containing an area of hemorrhage and yellow foci of necrosis. Note these degenerative changes are not the norm, but may occur in larger seminomas.
The stroma is usually in the form of thin fibrous strands that separate the neoplastic cells into lobules. Occasionally, the septa may be broad and hyalinized. The septae usually contain varying amounts of lymphocytes and in up to 50% of cases, granulomas.
Classic fried egg appearance can be appreciated --the neoplastic cell is large and round with clear cytoplasm and well-defined cell membranes. The centrally placed nucleus contains one or two prominent nucleoli. Note the presence of scattered lymmphocytes in the intervening fibrous stroma
The tumor contains adjacent intratubular germ call neoplasia (ITGCN) and associated intratubular granulomas. Note that granulomatous inflammation may also occur in the stroma and this finding is seen in up to 50% of cases.
Fibrous septae can vary in thickness as seen in this area. Mitotic figures are common. Scattered syncytiotrophoblasts are seen in 10-20% of cases and these patients may have elevated hCG, although not as elevated as patients with choriocarcinoma.1,2
IHC studies may be helpful, especially in instances when inflammation and granulomatous reactions obscures the tumor cells. The neoplastic cells will be immunoreactive with PLAP (placental alkaline phophatase).
And the cells are als reactive for C-kit (CD117) in a distinct membranous pattern. Antibodies against OCT4 (POU4F1), a transcription factor expressed in germ cells and embryonic stem cells, will also stain the nuclei of seminoma cells. Up to 40% of tumors will be focally positive for cytokeratins.1
Nuclear staining for OCT-4 is also a feature of seminoma.
D2-40 also shows diffuse strong staining but in a nice membranous pattern. D2-40 may also stain embryonal carcinoma, but the pattern tends to be focal and along the luminal aspect of the cells.
Seminomas are a common type of germ cell tumor (GCT), accounting for up to 50% of all testicular tumors. Note that when the term 'seminoma' is used, it is assumed that one is referring to the classic or usual type. The other seminoma is spermatocytic seminoma, which has entirely different clinicopathologic features. The ovarian counterpart of a seminoma is a dysgerminoma.
The most common cytogenetic aberration is an isochromosome 12p.
Mean age at diagnosis is between 35-45, which is 5-10 years later than patients with nonseminomatous GCTs. Clinical presentation is that of a painless testicular mass -- 10% may report scrotal pain. Elevated PLAP and hCG levels may be seen in some patients. An elevated AFP level is not typical of seminomas and should make one suspect a yolk sac tumor and should be treated as a nonseminomatous tumor.
Approximately 75% of seminomas are confined to the testes at the time of diagnosis, compared to 50-70% of nonseminomatous GCTs having metastasized at the time of diagnosis.2,3
Most seminomas present in Stage I (localized to the testis without metastasis), which is the paradigm of a curable malignancy. At the recent European Consensus Conference on Diagnosis and Treatment of Germ Cell Cancer, it was stated that all three treatment options following orchiectomy are acceptable --surveillance, adjuvant carboplatin, and adjuvant radiotherapy--are acceptable strategies for the management of patients with clinical stage I seminoma (Schmoll).
Five year survival rates according to stage are 99% (stage I), 89% (stage II) and 70-85% (stage III).1
• Testis : Spermatocytic Seminoma
• Testis : Intratubular Germ Cell Neoplasia
• Testis : Malignant Mixed Germ Cell Tumor
• Testis : Spermatocytic Seminoma
1 Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 816-820.
2 Kumar V, Abbas AK, Fausto N. Robbins and Cotran Pathologic Basis of Disease. 7th Ed. Philadelphia, PA: Elsevier; 2005: 1041-2.
3 Zhou M, Magi-Galluzzi, C. Genitourinary Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elvesier; 2006: 538-540.
Schmoll HJ,et al. ESMO Guidelines Working Group. Testicular seminoma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009 May;20 Suppl 4:83-8.