
This submucosal leiomyoma presents as a protruding well-circumscribed solid mesenchymal mass.
The bulging nodules are clearly distinct from the myometrium - note the whorled white-tan cut surface.
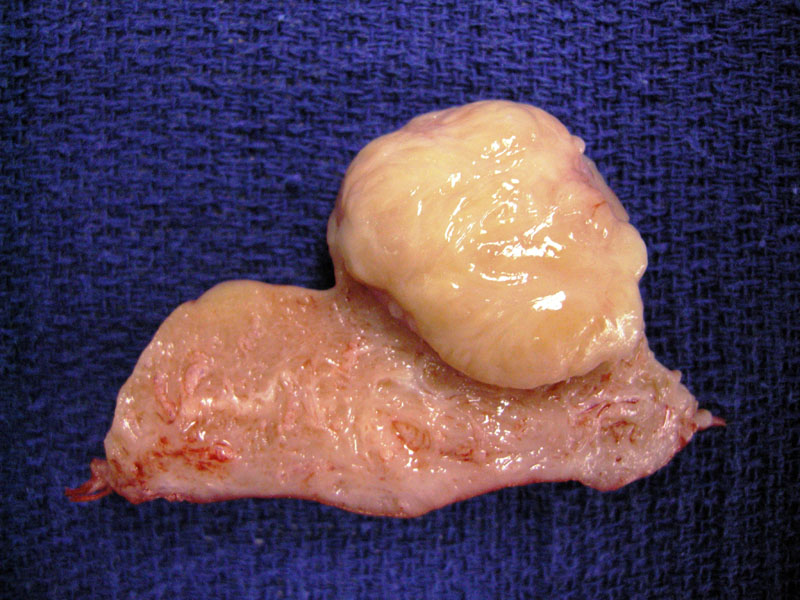
A large submucosal nodule (protruding toward you) is located endometrial cavity -- another intramural leiomyoma (top image) has been shelled out of the myometrium.
Microscopically, a conventional leiomyoma is composed of intersecting fascicles of spindle cells.
Here is a regular leiomyoma with secondary sclerosis. Degenerative changes can occur, especially in larger leiomyomas.
Hemorrhage is a common degenerative feature as well.
This image demonstrates a poorly demarcated zone of eosinophilic detritus due to ischemic necrosis. This differs from the sharply defined border of true tumor necrosis, which is a feature of leiomyosarcoma.
Leiomyoma with hydropic degeneration due to accumulation of edema fluid. The hydropic regions may be focal, or as in this leiomyoma, more diffuse.
Another variant is the cellular leiomyoma -- the spindle cells are closely spaced and there is no mitotic activity. The definition is fairly vague, but most pathologists can recognize hypercellularity since we see so many usual-type leiomyomas.
Scattered embedded adipocytes are seen in this lipoleiomyoma -- this benign variant usually arises in older, post-menopausal women compared with typical leiomyoma. Abnormalities in chromosome 5 have been detected -- these benign often appears more yellow grossly due to the fat content.
Here is quite an unusual variant, a myxoid leiomyoma with hematopoietic elements. Myxoid blue material can be seen some cystic spaces (upper left) and the central space contains an infiltrate of lymphocytes and maybe plasma cells and eosinophils. The etiology of these cells is unclear, and it is uncertain if true extramedullary hematopoiesis is taking place.
Leiomyomata, also known as fibroids, are the most common solid tumors in women. They are also the most common indication for a hysterectomy. These benign tumors of the muscular uterine wall are clonal neoplasms which exhibit wide variety of cytogenetic aberrations.
Grossly, these tumors are sharply circumscribed, round, firm, tan-white masses that bulge out from the cut uterine surface. In fact, they can almost be 'shelled-out' from the surrounding myometrium. Leiomyomata can be located within the myometrium (intramural), just underneath the endometrium (submucosal) or just beneath the serosa (subserosal). Degenerative changes such as hemorrhage, cysts, ulceration and edema may occur, especially in larger leiomyomata or submucosal leiomyomata.1,2
Numerous variants of leiomyomata exist and they are beyond the scope of this discussion. These variants can be quite unusual or atypical, but they are usually benign actors. Of course, the pathologist must be familiar with criteria that separate the leiomyomata and its benign variants from the dreaded leiomyosarcomas, which are highly malignant sarcomas.
Leiomyosarcomas exhibit increased mitotic activity, nuclear atypia and coagulative necrosis. As you might have guessed, the criteria for separating mitotically active, atypical and cellular variants of leiomyomas from leiomyosarcomas is often not clear cut. There is currently debate regarding exactly how mitotically active, atypical or necrotic a tumor needs to be to be labeled a leiomyosarcoma. This is why a category "smooth muscle tumor of unknown malignant potential (STUMP)" was created -- to have a name for these nebulous smooth muscle tumors that stump pathologists, so to speak!
Briefly, the leiomyoma subtypes include: cellular, highly cellular, mitotically active, hydropic, atypical, leiomyoma with secondary hormonal changes, plexiform, epithelioid, myxoid and lipoleiomyoma. Unusual growth patterns include: intravascular leiomyomata, disseminated peritoneal leiomyomatosis and benign metastasizing leiomyoma.1-3
These tumors occur in reproductive age women and estimated frequency in hysterectomy specimens approaches 77%. Only a subset of these tumors are symptomatic, and symptoms correlate with tumor size and location. For example, submucosal leiomyomas may cause abnormal uterine bleeding. Uterine enlargement may result in abdominal fullness, pelvic pain, menorrhagia, hydronephrosis from ureteral compression, and urinary frequency from compression of the bladder. Infertility and spontaneous abortion may result from impaired blastocyst implanation secondary to a submucosal leiomyoma and/or a severely distorted uterine cavity.1,2
Black women tend to have leiomyomata at a younger age and also, multiple and larger leiomyomata. Nulliparous women also have an increased risk of these neoplasms. Leiomyomata express estrogen and progesterone receptors and are thus hormonally responsive. They undergo rapid growth during pregnancy (or clomiphene therapy) and regress after menopause. They also exhibit increased mitotic activity during the secretory phase of the menstrual cycle.
Treatment depends on the symptoms a patient is suffering. Gonadotropin releasing hormone agonists may induce a chemical menopause that can result in decreased menstruation and decreased uterine volume. Abnormal uterine bleeding can be regulated with a variety of hormonal therapies. Surgically leiomyomata may be removed via myomectomy or hysterectomy for definitive therapy. If intracavitary lesions are present they may be removed hysteroscopically. More recently, uterine artery embolizations have been employed to for patients wishing to avoid the operating room.
Rapidly growing leiomyomas may raise concerning for malignant transformation to leiomyomsarcomas, however, statistically speaking, the risk of malignant transformation is quite low.
• Myometrium : Leiomyoma, Mitotically Active
• Myometrium : Leiomyoma, Highly Cellular
• Myometrium : Intravascular Leiomyomatosis
• Myometrium : Leiomyosarcoma, Epithelioid Variant
1 Kumar V, Abbas AK, Fausto N. Robbins and Cotran Pathologic Basis of Disease. 7th Ed. Philadelphia, PA: Elsevier; 2005: 1089-90.
2 Nucci MR, Oliva Esther. Gynecologic Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elsevier: 2009: 261-278.
3 Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 683-688.