
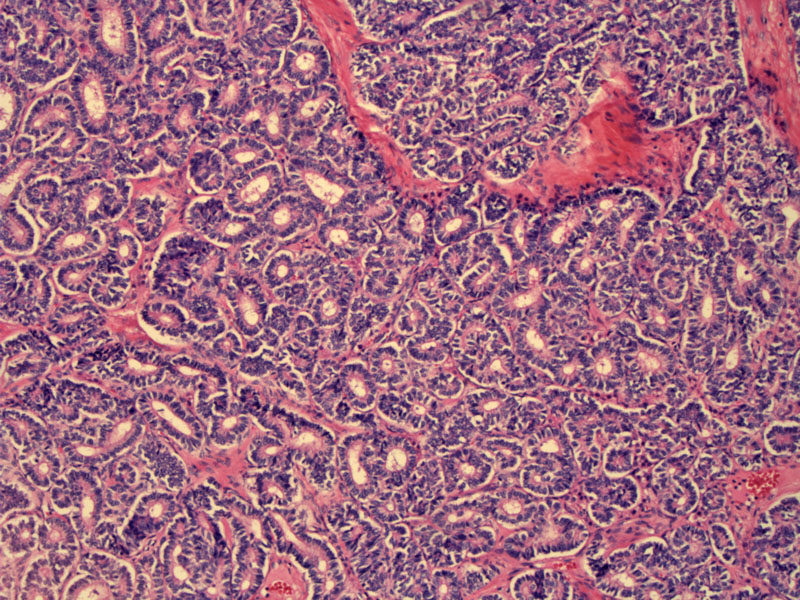
Well-differentiated SLCT consists of solid or hollow tubules embedded in a fibrous stroma.
The tubules are lined by mature Sertoli cells, which have abundant eosinophilic cytoplasm and round basally oriented nuclei. The cytoplasm may also be pale and slightly vacuolated due to lipid content. Note that in the left lower corner, there are several Leydig cells with round nuclei and pink cytoplasm that blends in with the stroma.
This intermediately differentiated SLCT consists of trabeculae of less mature Sertoli cells with hyperchromatic, mildly pleomorphic nuclei and scant cytoplasm. Leydig cells are not seen in the fibrous stroma. One would have better luck looking for Leydigs are the periphery of the tumor.
Other areas of the above tumor demonstrated cords of tumor cells separated by an edematous stroma, a very common pattern for intermediately differentiated SLCT.
Yet other areas exhibited compression of cords to form more solid areas.
A different case of well to intermediately differentiated SLCT exhibits a nested pattern. Although the malignant Sertoli cells are still fairly well-differentiated with basally oriented nuclei and abundant cytoplasm, the overall picture is more jumbled and crowded. The fine cracking seen within the tubules may be artifactual or due to tumor cell dehiscence.
Another area of the above tumor demonstrates tubules of malignant Sertoli cells embedded within a fibrous stroma.
A retiform SLCT demonstrates an irregular network like proliferation of tubules and cysts. Papillae may project into the cysts, as seen on the right. The tubules and cysts are lined by cuboidal cells with
Poorly-differentiated SLCT consists of a proliferation of round to spindled cells, resembling a sarcoma.
Mitotic figures are frequently found in the poorly-differentiated variant.
A different case demonstrates an admixture of Sertoli cells (dark oblong nuclei with a small rim of cytoplasm) and Leydig cells (round nuclei with pale abundant pinke cytoplasm).
A well-differentiated area consisting of well-formed variably-sized tubules of Sertoli cells can be seen.
Areas of solid growth consisting of lipid-rich Sertoli cells can be appreciated. The cytoplasm of Sertoli cells can be pale to eosinophilic (depending on the amount of lipid).
The cystic lumens may be filled with eosinophilic secretions, bearing a resemblance to thyroid follicles.
Yet another view of Sertoli cells intimately mixed with Leydig cells arranged in a diffuse solid pattern. Although many intermediately differentiated SLCTs have a lobulated architecture on low power, solid areas are not uncommon.
Much of this tumor was arranged in a trabecular growth pattern, resembling a neuroendocrine neoplasm.
Inhibin is also a useful marker but also stains other sex cord stromal tumors such as granulosa cell tumors.
These tumors often show focal positivity for MelA-103 as seen here by the red staining of some cellular cytoplasm.
Sertoli-Leydig cell tumor (SLCT) can be divided into five histologic categories:1
(1) Well-differentiated: These tumors comprise 10% of cases and are composed of solid or hollow tubules lined by cuboidal or columnar Sertoli cells, surrounded by a fibrous stroma which contains Leydig cells. The Sertoli cells contain abundant eosinophilic cytoplasm with basally oriented round or ovoid nuclei. These tumors almost never contain retiform or heterologous elements.
(2) Intermediately differentiated: Cellular proliferation of Sertoli cells (arranged in diffuse nodular sheets, poorly formed tubules, nests or trabeculae) are separated by a hypocellular edematous stroma, imparting a lobulated appearance at low power. A diffuse solid arrangement can also be seen. The Sertoli cells are less "mature" with scant cytoplasm. Leydig cells may be admixed with the Sertoli cells, although they are most easily identified in the periphery of lobules, embedded in the edematous stroma.
(3) Poorly differentiated: Sarcomatoid growth of poorly-differentiated Sertoli cells is seen, resembling a sarcoma. The Sertoli cells are barely recognizable, with scant cytoplasm and hyperchromatic nuclei. Mitotic activity is brisk. Poorly formed nests and tubules of Sertoli cells and scant Leydig cells are seen focally. Note that there is a discrepancy between several authors regarding the origin of the sarcomatous component of this subtype. Fletcher describes the sarcomatous component as immature stromal cells, whereas Nucci states they are actually poorly-differential Sertoli cells. Neither Sternberg nor Rosai clarify this point.
(4) Retiform variant: Retiform (defn: net-like) histology is seen in approximately 15% of intermediate and poorly differentiated SLCTs. The growth pattern recapitulates that of the rete testis, with an irregularly branching network of slit-like tubules and cysts. The cysts may contain papillary structures.
(5) Heterologous elements: Seen in intermediate or poorly-differentiated tumors, the most common heterologous elements is mucinous gastrointestinal epithelium. These glands and cysts generally elaborate deeply eosinophilic secretions. Mesenchymal heterologous elements include immature cartilege and skeletal muscle.
If either retiform or heterologous elements are identified, this must be mentioned in the pathology report as it connotes a worse prognosis.
Typically occurs in young women and 75% of SLCTs are diagnosed before age 30. Interestingly, well-differentiated SLCTs occur 10-15 years later, and the average age is 45. Retiform SLCTs occur occur 10-15 years earlier, with the average age of 15-16 at presentation.1,2
One-third to one-half of tumors secrete enough androgens to cause virilization in patients. Serum levels of testosterone and urine 17-ketosteroids are elevated. Nonspecific symptoms include abdominal pain and distention.1,2 The vast majority of tumors are unilateral. Well-differentiated tumors average 5 cm in diameter, while intermediate and poorly-differentiated tumors are larger and average 15 cm in diameter. Grossly, the tumor is solid yellow to tan cut surface with focal cyst formation. Necrosis and hemorrhage are common secondary changes in poorly-differentiated tumors.
Generally excellent. Stage, degree of differentiated and presence of retiform or heterologous elements are important prognostic factors. Well-differentiated tumors are clinically benign. Approximately 80% of patients present with stage IA tumors, 15% have stage IC tumors (peritoneal washings have positive malignant cells due to tumor rupture) and 5% have extraovarian spread.
5-year survival rate is 92% and recurrence after excision occurs in 18% of cases in recent studies. One large study (Zhang) found younger age and early-stage disease are important predictors for improved survival in patients with ovarian sex cord stromal tumors. This age-associated survival advantage was present for early, but not advanced-staged patients. Similarly, another study (Chan) found on multivariant analysis, age <50 (P = 0.003), premenopausal status (P = 0.013), tumor size < 10 cm (P = 0.003), lack of lymph node invasion (P < 0.0005), and absence of residual disease (P = 0.002) to be significant predictors for improved survival (Chan).
• Ovary : Endometrioid Adenocarcinoma, Sertoliform Pattern
1 Nucci MR, Oliva Esther. Gynecologic Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elsevier: 2009: 473-480.
2 Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 595-8.
Zhang M et al. Prognostic factors responsible for survival in sex cord stromal tumors of the ovary--an analysis of 376 women. Gynecol Oncol. 2007 Feb;104(2):396-400. Epub 2006 Oct 9.
Chan JK, et al. Prognostic factors responsible for survival in sex cord stromal tumors of the ovary--a multivariate analysis.Gynecol Oncol. 2005 Jan;96(1):204-9.