Histologic staging scheme for mucoepidermoid carcinoma.
FNA shows a cellular smear consisting of cohesive sheets of epithelial cells with a mucinous background. The cells may be intermediate cells, mucocytes and epidermoid cells composing the tumor. Usually, the cells will have a lot of cytoplasm and little pleomorphism unless the tumor is high-grade.
Another FNA showing cohesive sheets of (most likely) intermediate cells. It is difficult to see the mucocytes with intracytoplasmic mucin vacuoles
Composed of mainly 3 cell types (epidermoid cells, intermediate cells and mucocytes). Other less common cell types are clear cells, columnar cells and oncocytes. Usually, the intermediate cell type predominates, as seen here. Intermediate cells can look
Mucocytes often line cysts and ductal spaces of the tumor. Here, scattered mucocytes are dispersed among the intermediate cells.
Focal clear cell change is common. Clear cells have centrally placed nuclei (vs mucocytes with eccentrically located nuclear) and clear cytoplasm (vs mucocyte with frothy, foamy cytoplasm).
Mucicarmine stain will highlight the intracytoplasmic mucin vacuoles inside the mucocytes
Mucoepidermoid carcinoma accounts for 5% of all salivary gland tumors and 20% of the malignant ones, and is the most frequent primary malignancy of the salivary gland in both adults and children.
A subset have been show to have a recurring chromosomal translocation, t(11;19)(q21;p13), which is often the sole cytogenetic alteration (El-Nagger). The resulting CTCR1–MAML2 fusion has been detected in up to 40% of primary salivary gland mucoepidermoid carcinomas, and the CRCR-3-MAML2 fusion ones (found in up to 6%) are associated with a distinct tumor subset with favorable clinicopathological features including younger age, and an indolent clinical course (Nakayama; Okabe).
Usually occurs between the third and sixth decade of life, but it may occur at any age, with a predilection for women (F:M ratio is 3:2). Most common malignant salivary gland tumor, with most involving the parotid gland. Can also involve the palate, buccal mucosa, lips, upper and lower respiratory tracts. Also the most common malignant salivary gland tumor in children. Commonly presents as a painless, fixed, slow-growing mass, but may have an accelerated growth phase and other symptoms of pain, nerve involvement depending on location and grade of tumor.
Excision with negative margins (with or without neck dissection of lymph nodes) with adjunctive radiation for high grade tumors.
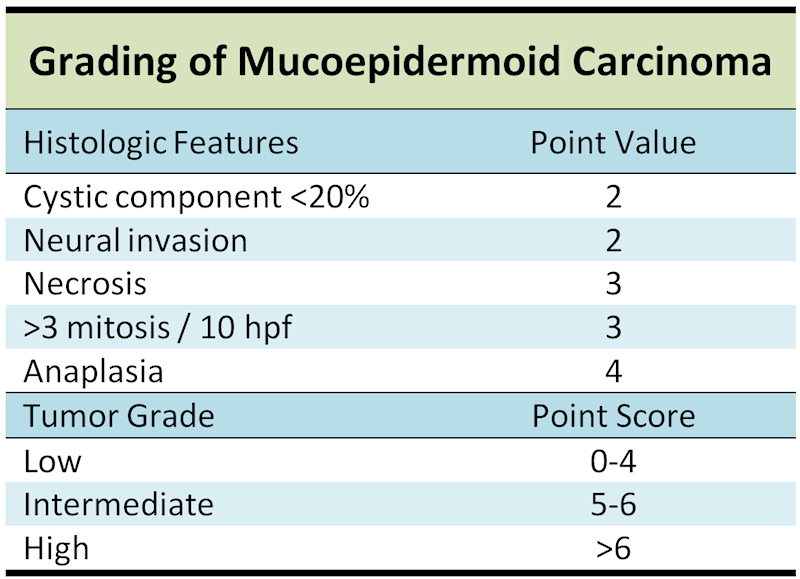
Depends on site of tumor, stage and grade and adequancy of resection. 40% recur locally. 15% will metastasize. Grading score (0-14) gives point values for the following morphologic features: intracystic component less than 20%, neural invasion, necrosis, mitotic figures and anaplasia. Mortality rate for low grade is 3.3%, intermediate grade is 9.7% and 46.3% for high grade .
Also of interest is that the presence of the MECT1-MAML2 fusion transcript has been reported to be associated with a less advanced clinical stage, longer disease-free and overall survivals on univariate analysis and in one study at least, emerged as an independent prognostic factor for longer overall survival on multivariate analysis (Okabe).
El-Naggar AK, Lovell M, Killary AM, et al. A mucoepidermoid carcinoma of minor salivary gland with t(11;19)(q21;p13.1) as the only karyotypic abnormality. Cancer Genet Cytogenet 1996;87:29–33.
Nakayama T, Miyabe S, Okabe M, Sakuma H, Ijichi K, Hasegawa Y, Nagatsuka H, Shimozato K, Inagaki H. Clinicopathological significance of the CRTC3-MAML2 fusion transcript in mucoepidermoid carcinoma. Mod Pathol. 2009 Dec;22(12):1575-81.
Okabe M, Miyabe S, Nagatsuka H, et al. MECT1-MAML2 fusion transcript defines a favorable subset of mucoepidermoid carcinoma. Clin Cancer Res 2006;12:3902–3907.
Behboudi A, Enlund F, Winnes M, et al. Molecular classification of mucoepidermoid carcinomas-prognostic significance of the MECT1-MAML2 fusion oncogene. Genes Chromosomes Cancer 2006;45:470–481.