
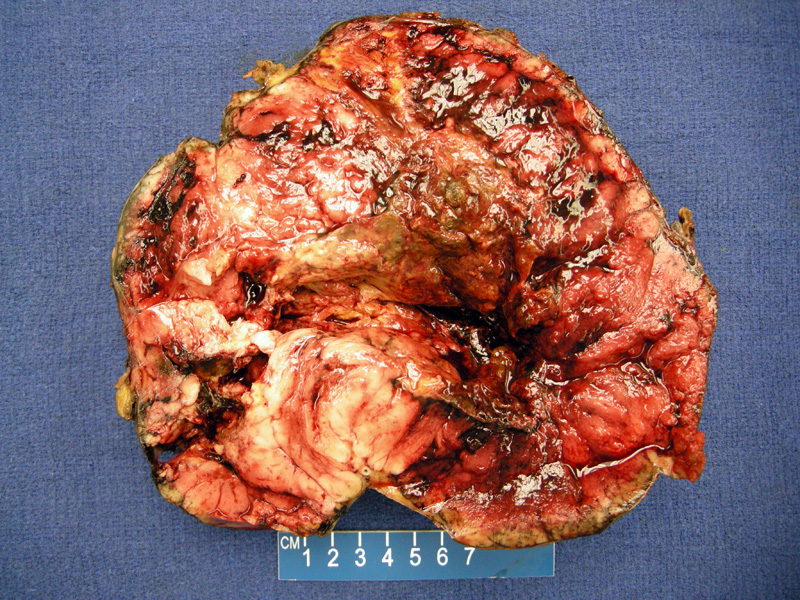
A large duodenal GIST with extensive hemorrhage and necrosis is seen here. It is difficult to identify the tumor mass, but it you can see a solid tan fleshy area at the lower left pole.
Most duodenal GISTs are composed of spindled cells such as seen here. This tumor contains blunted spindled cells which appear relatively bland.
Skenoid fibers (pink sclerotic areas) are known to occur in small intestinal GISTs. These extracellular aggregates of collagen can be highlighted by PAS stain and are considered a favorable feature.
Cellular pallisading is well-recognized in small intestinal GISTs as seen here by the nuclei lining up similar to a neural tumor. This feature can also be seen in the gastric GISTs, however.
This spindle cell area shows an increase in cellularity.
Coagulation necrosis is seen in some duodenal GISTS, most of which are large tumors, >5 cm.
C-kit is diffusely positive as expected.
CD34 stains the normal vascular structures but is lacking in the tumor cells. Only about half of duodenal GISTs have been shown to stain for CD34 (Miettinen). About 20% of these tumors show focal S-100 staining (Miettinen).
A Diff-Quick stain of a tumor FNA shows the same bland spindle cells with some stromal elements. Note again the nuclear pallisading seen in the histological sections.
The pap stained smear shows a cellular spindle cell lesion with loose cohesion.
GISTs arise more often in the stomach (60%) and small bowel (35%). Less than 5% occur in the esophagus, omentum and mesentery. About 5% of GISTs occur as part of an inherited syndrome, either NF1 or Carney triad (gastric epithelioid GISTs, pulmonary chondroma and extra-adrenal paragangliomas). In these familial GISTs, there is an germline mutation of either KIT or PDGFRA.
Gastric GISTs display two major morphologies, spindled and epithelioid. PDGFRA mutations appear to be more common in the epithelioid subtype and this particular mutation connotes an improved prognosis (Fletcher, Miettinen 2006).
However, in the duodenum and the rest of the small intestines, there are no distinct histologic subtypes and the vast majority are composed of spindle cells. About half of small intestinal GISTs exhibit skenoid fibers, which are PAS positive aggregates of extracellular collagen. Skenoid fibers are favorable features.
In contrast to gastric GISTs, epithelioid morphology in small intestinal GISTs are associated with a more malignant tumor. Miettinen states "small intestinal epithelioid GISTs probably represent a morphologic manifestation of tumor progression rather than a distinct histologic subtype" (Miettinen 2006).
I am not aware of a study detailing PDGFRA mutations in the duodenum, but Miettinen, in a separate study, conducted a study of 906 jejunal and ileal GISTs (excluding the duodenum) and did not find PDGFRA mutations of exon 18 or 12 -- the majority exhibited mutations of exon 11 in the KIT gene, which is the most frequently mutated exon in gastric GISTs (Miettinen 2006b).
While duodenal GISTs are less frequent and account for <5% of all GISTs, they still represent approximately 30% of primary duodenal tumors (Miettinen 2003). Other differential diagnoses to consider would include leiomyomas and leiomyosarcomas.
Similar to GISTs of other sites, duodenal GISTS affect primarily older adults with median age of 55-60 years (Miettinen 2006). These tumors have also been found in patients <40 years (including a pediatric example) slightly more often than GISTs of other locations (Miettinen 2003).
The clinical presentation of duodenal GIST varies according to their size and whether there is mucosal ulceration, and commonly includes gastrointestinal bleeding with chronic anemia or abdominal pain, while a substantial number are detected incidentally. Some cases present with a palpable mass or intestinal obstruction.
The optimal management of GISTs remains surgical resection with clear margins. Because lymphatic spread is rare, nodal dissection is not required. Wedge resection seems sufficient for small lesions if the ampulla of Vater can be preserved, while segmental duodenectomy is necessary for larger tumors located at the third and fourth portion of the duodenum. Partial duodenectomy with Roux-en-Y duodenojejunostomy is recommended for larger tumors involving the antimesenteric border of the second and third portion of the duodenum (Chung).
The use of imatinib mesylate, a competitive inhibitor of KIT, is effective in most patients with advanced or metastatic GISTs.
Similar to GISTs at other sites, the spectrum of duodenal GISTS range from clinically benign to overt sarcomas -- the two most important parameters are size and mitotic activity. Note, however, these parameters must be interpreted in the context of the inherently higher malignant potential of small intestinal GISTs (Miettinen 2006). It is not known why small intestinal GISTs of the same size and mitotic activity as a gastric GIST are more likely to invade and metastasize.
One study of 156 duodenal GISTS (Miettinen 2003) concluded the following:
(1) Small tumors <2 cm in diameter and with <=5 mitoses/50 HPF seem to be indolent, with no detectable risk for recurrence or metastases.
(2) Tumors >2 cm but not >5 cm with a low mitotic rate showed a low frequency of malignant behavior. Three (comprising 6% of this group) patients died of tumor after a prolonged course of disease (over 5 years).
(3) Tumors with >5 mitoses/50 HPF behave in a malignant fashion. Tumors >5 cm seem even more malignant because 86% of the patients died of disease, whereas the tumor-related mortality was 50% for such tumors <5 cm. Metastases most commonly involve intraabdominal soft tissues and liver.
• Small Intestines : PDGF-R Related Gastrointestinal Stromal Tumor
• Stomach : Gastrointestinal Stromal Tumor (GIST)
Chung JC, et al. Management and outcome of gastrointestinal stromal tumors of the duodenum. J Gastrointest Surg. 2010 May;14(5):880-3.
Miettinen M, et al. Gastrointestinal stromal tumors, intramural leiomyomas, and leiomyosarcomas in the duodenum: a clinicopathologic, immunohistochemical, and molecular genetic study of 167 cases. Am J Surg Pathol. 2003 May;27(5):625-41.
Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006 Oct;130(10):1466-78.
Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006 Apr;30(4):477-89.