
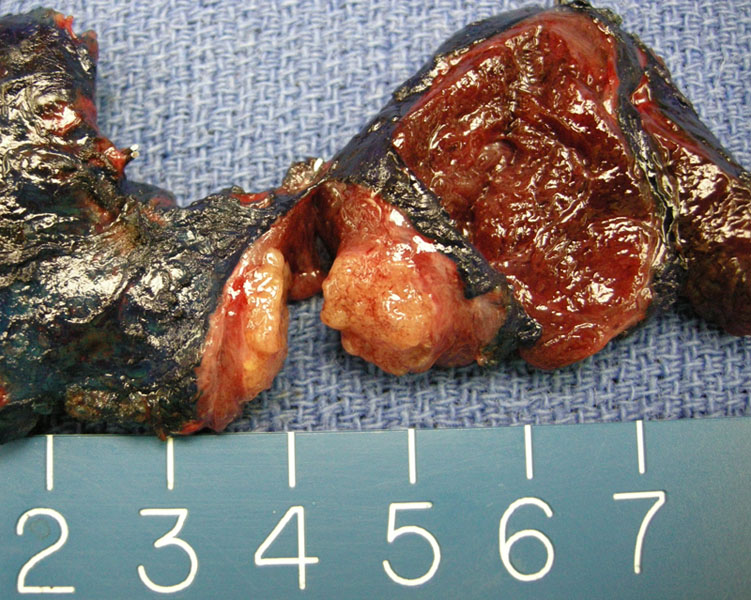
Papillary carcinoma is bulging from the cut surface as a tan lobulated solid mass.
FNA shows crowded follicular cells in a papillary architecture, which makes the diagnosis relatively straight-forward. Now a search for nuclear abnormalities is in order.
Other lesions may only demonstrate small sheets of small irregular sheets without any overt papillary features.
Nuclear clearing may be appreciated sometimes in the diff-quick preparation.
Areas with twisted sheets are characteristic of papillary carcinoma.
A nice intranuclear inclusion can be appreciated in this cluster (arrow).
The conventional type of papillary carcinoma shows many well-developed papillary structures.
The cyst wall shows attenuated epithelium but close inspection reveals some cells with intranuclear inclusions as well as irregularity.
Other areas show back-to-back macrocysts lined by hobnailed cells.
Papillary carcinoma may have dense stromal calcifications as well.
Infarction of papillary tips may result in psammomatous calcifications.
Occasionally there may be extensive osseous metaplasia (on the left) associated with a conventional papillary thyroid carcinoma.
Another FNA of a different papillary carcinoma shows high cellularity consisting of irregular groups, some of which overlie one another.
Another example shows classic papillary carcinoma with areas of macrocystic architecture.
This is an example of papillary-like infoldings in a colloid nodule. Note the lack of nuclear features of papillary carcinoma. Beware -- this is not papillary thyroid carcinoma.
Papillary thyroid cancer (PTC) is the most common malignancy of the thyroid gland, accounting for about 80% of all primary thyroid malignancies. It is about three times as common in women, however, the risk of death from PTC is twice as high for males (Hundahl).
Papillary thyroid carinoma is characterized by follicular differentiation and distinctive nuclear features. Parenthetically, follicular carcinomas of the thyroid also demonstrate follicular differentiation but do NOT exhibit the distinctive nuclear features seen in PTC. Thus, the nuclear features of PTC should be well-ingrained in your mind as it will help you decide what neoplasm falls under the papillary category and what falls under the follicular category. This will especially important in confusing entities such as the follicular variant of PTC or follicular adenoma with papillary features.
Grossly, PTC typically exhibits an infiltrative border with a firm consistency and can be multifocal in more than half the cases. Less commonly, the tumor may be encapsulated. The cut surface is granular due to numerous papillae. In some tumors, actually papillary can be discerned. PTC with more follicular architecture exhibit a more fleshy cut surface. Often, the tumor can feel gritty due to presence of psammoma bodies (Fletcher).
The nuclear features of PTC:
- enlarged and overlapping nuclei with fine, powdery, "ground class" or "Orphan Annie" appearance
- intranuclear cytoplasmic inclusions
- nuclear grooves with irregular nuclear outlines
You often here about "Orphan Annie eyes", and this refers is an old cartoon depiction of Orphan Anne where she does not have pupils, but just a white space for her eyes. A bit freaky if you ask me. The neoplastic cells can be polygonal, cuboidal, columnar or hob-nailed and the cytoplasm ranges from eosinophilic to clear. The papillary structure is usually evident. Note that in metastatic lymph nodes, cystic change is common with a loss of the usual PTC nuclear features, thus, it can easily be misdiagnosed as a cyst or another benign structure (Fletcher).
Molecular Markers.
The molecular changes underlying papillary carcinoma have been extensively studied and play an important role in understanding the pathogenesis and the clinicopathologic features of the disease.
RET/PTC Rearrangements: RET is a proto-oncogene that encodes a protein tyrosine kinase receptor with an extracellular domain, a transmembrane domain, and an intracytoplasmic kinase domain. RET is normally expressed in cells derived from the neural crest and in the kidney. However, RET is involved in a number of rearrangements known as RET/PTC that have been implicated in the tumorigenic process of PTC. There are a number of RET/PTC gene rearrangements, of which RET/PTC1 and RET/PTC3 are the most common. These RET rearrangements are restricted to the thyroid gland and are fairly specific for PTC. The RET/PTC rearrangements are considered to be early events in tumorigenesis, present even at high frequency in microcarcinomas.
BRAF Mutations. BRAF is a protein kinase that has an important role in cell proliferation, differentiation, and programmed cell death. Activating mutations of BRAF were found initially in human colon cancers and malignant melanoma. It is now known that BRAF is also mutated in PTC with high frequency. The most common mutation is characterized by change of valine to glutamate in codon 600 (V600E), leading to increased kinase activity. The BRAF mutations are associated most often with the classic variant and, because these usually are not diagnostic dilemmas, they usually do not require this ancillary technology, but it can be helpful for nondiagnostic cytology specimens.
Patients with PTC typically present with a thyroid mass, found on exam or other screening tests such as ultrasound. Workup includes FNA and imaging studies. Iodinated contrast agent is contra-indicated as it interferes with post-operative I131 ablation.
Additionally some patients may present with lymph node metastasis and upon further investigation, an occult primary is found on the ipsilateral side (Fletcher). In higher stage tumors, PTC can invade locally into the thyroid parenchyma and perithyroid soft tissue. Metastasis usually goes to regional lymph nodes. Distant metastasis is uncommon and a late manifestation of disease (Fletcher).
Patients can be classified into low-risk and high-risk groups. The low-risk patients includes patients under the age of 45 with primary lesions under 2 cm and no evidence of intra- or extraglandular spread.
Treatment for confirmed PTC is removal of the entire thyroid gland. With solitary micro lesions <3 mm there is probably a role for observation after conservative surgery(with consideration of post-surgical suppression), however, the mainstay of surgical treatment is total thyroidectomy.
A complete thyroidectomy facilitates post-operative radioactive iodine ablation of any remnant malignant disease. Normal thyroid tissue is more iodine avid than neoplastic disease and large doses of I131 would be needed to ablate normal residual thyroid tissue. Additional arguments for total thyroidectomy are the high risk of multi-centric disease and the use of thyroglobulin as a tumor marker postoperatively.
Management of the lymph nodes is evolving. In the past a wait and see approach was used and when patients had clinically evident metastatic lyphadenopathy containing PTC, a "berry-picking" strategy was employed to remove positive nodes. At this time, most authors recommend the removal of whole levels of lymph nodes rather than the single node-ectomy. Management of the N0 neck is more controversial with some authors advocating routine paratracheal lymphadenectomy(1st echelon nodes) while others reserve this procedure for higher risk tumors based on various staging systems. Because PTC has such a high propensity for metastasis, elective central neck dissection is becoming incorporated in standard treatment paradigms.
Generally, the prognosis is excellent with usually ~95% ten year survival (Hundahl). There are numerous proposed systems to stratify patients based upon various factors. Some of the more popular systems for stratifying prognosis include: AGES (Age, Grade, Extent of disease, Size), AMES (Age, Metastasis, Extent of disease, Size), and TNM (Tumor size, Regional Nodes, Distant Metastasis). For well differentiated PTC, research has historically needed large patient populations to detect differences in outcomes. This is because the overall the prognosis from PTC is excellent in most instances.
→PTC is defined as a thyroid tumor with follicular cell differentiation and distinct nuclear features.
→The nuclear features of PTC include crow\ded nuclei with a clear-out ground-glass or "Orphan Annie eye" appearance, nuclear grooves, distinct nucleoli and nuclear pseudoinclusions.
• Thyroid : Papillary Carcinoma, Columnar Variant
• Thyroid : Papillary Carcinoma, Follicular Variant
• Thyroid : Papillary Carcinoma, Solid Variant
• Thyroid : Papillary Carcinoma, Tall Cell Variant
• Thyroid : Papillary Carcinoma, Warthin's Variant
• Thyroid : Papillary Thyroid Carcinoma, Cribriform-Morular Type
• Thyroid : Hyalinizing Trabecular Adenoma
Hundahl SA, Fleming ID, Fremgen AM, Menck HR. A national cancer data base report on 53,856 cases of thyroid carcinoma treated in the US, 1985-1995. Cancer 1998;83:2638-48.
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 1000-6.